Medical network - May 2, to fully reflect the situation of adverse drug reactions monitoring in China in 2016, strengthen the supervision of drugs and to carry out the enterprise's main body responsibility, promote the clinical rational drug use, and ensure the public safety, on the basis of the adverse drug reaction report and monitoring measures for the administration, the state food and drug supervision and administration bureau (hereinafter referred to as the bureau) organize national adverse drug reaction monitoring center compiled the national adverse drug reaction monitoring annual report (2016).
First, the progress of the monitoring of adverse drug reactions
The network construction continued to deepen and the monitoring capability was further enhanced. In 2016, the national drug adverse reaction monitoring network has been further deepened, and our country's ability to detect and collect information about adverse reactions in medicines has been further enhanced. Rapid growth of network users at the grass-roots level, the country has more than 310000 medical institutions, pharmaceutical production enterprise registration for adverse drug reaction monitoring network users, and through the network to submit the report adverse drug reactions. The country's 97.7 percent of counties reported adverse drug reactions, with an average report of 1,068 per million people, up from 2015.
Analytical evaluation capability continues to be strengthened, and risk management and communication are more open. In 2016, the report and monitoring of adverse reactions were conducted in an orderly manner. Through monitoring day, weekly summary, quarterly analysis method such as strengthen the national adverse drug reaction monitoring data analysis and evaluation, to dig deeper into drug risk signal, the including testosterone drugs, phenacetin drugs, monosodium phosphate for injection, vidarabine, compound leaf tablet, compound amino acid injection (18 aa) and similar preparations, a fairy bone remains of oral preparations such as 42 (class) varieties was carried out safety evaluation, and adopt corresponding measures for risk management and communication. According to the result of the safety evaluation, the Suggestions are proposed for 35. Iv. The "drug alert express" 12, a total of 63 information, involved 68 varieties; The announcement of the release of two varieties of chlormethadone and benzoguanidine was announced.
The monitoring of adverse events will continue to be enhanced and the effectiveness of the warning will be improved. Specification adverse drug events clustering signal disposal, to strengthen the construction of early warning platform, increase the efficiency of the early warning work, to focus on more than 150 adverse drug events clustering signals in a timely manner disposal, confirmed by the evaluation of progesterone injection six gather event risk control measures, such as further and ensure public safety.
The responsibility of enterprises will continue to be implemented and the level of drug safety and safety will be further enhanced. In 2016, we will strengthen training in the monitoring of the monitoring of adverse reactions of pharmaceutical companies, promote the awareness of the responsibility of drug manufacturers and improve their ability to evaluate drug safety. Monthly feedback adverse reactions monitoring data to pharmaceutical production enterprises, guide enterprises to carry out data analysis and evaluation, take timely measures to control risk, and implement the responsibilities for enterprise security risk main body.
Adverse reactions/incident reports of drugs
(1) the overall situation of the report
The 2016 drug adverse reaction/incident report
In 2016, the national drug response monitoring network received 143, 000 drug adverse reactions/incident reports, up 2.3 percent from 2015. From 1999 to 2016, the national drug adverse reaction monitoring network received nearly 10.750,000 copies of "adverse reactions/events report".
FIG. 1. The growth in the number of adverse drug reactions/events reported in China in 1999-2016
New and serious adverse reactions/incident reports
In 2016, the national drug response monitoring network received a new and serious adverse reaction/incident report of 42.3 million, up 7.4 percent from 2015. New and serious reports accounted for 29.6% of the total, up by 1.4 percentage points from 2015. The proportion of new and severe adverse reactions/events reported continued to increase, showing that the overall availability of drug adverse reactions in China was increasing.
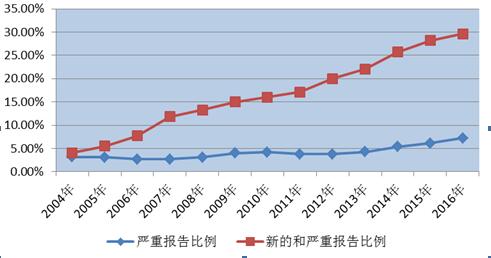 FIG. 2 - new and severe and severe drug adverse reactions/event reporting ratios in 2004 and 2016
The average case report for every million people
The number of reported cases per million people is one of the most important indicators of the level of monitoring of adverse drug reactions in a country. In 2016, the average number of reported cases per million people in China was 1,068, an increase of 2.4 percent from 2015.
The proportion of adverse reactions/events at the county level?
The proportion of adverse drug reactions/events at the county level is one of the important measures to measure the balanced development and coverage of adverse drug reactions in China. The number of national drug adverse reactions/events in 2016 was 97.7 percent, an increase of 1.1 percentage points from 2015.
Drug adverse reactions/event reporting sources
Drug production enterprises, business enterprises and medical institutions are responsible units for the report of adverse drug reactions. The 2016 drug adverse reaction/incident report was based on the source, accounting for 85.6 percent of the report from the medical establishment; Reports from pharmaceutical companies account for 12.8 percent. Reports from drug manufacturers accounted for 1.4%; Personal and other reports account for 0.2%.
 FIG. 3: the distribution of adverse drug reactions/event reports in 2016
The reporter's job
According to the report's career statistics, the doctors reported 55.5%, the pharmacists reported 25.3%, nurses reported 15.1%, and other reports accounted for 4.1%. The report on the 2015 report is basically the same.
Figure 4. The speaker's career composition
The adverse reaction/incident report of the drug involves the patient
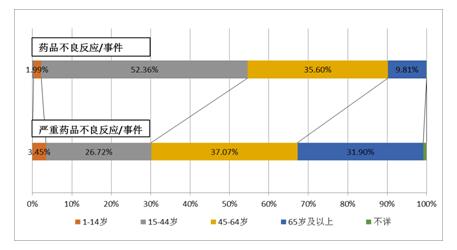
The proportion of men and women was close to 0.89:1, slightly more women than men, and gender distribution trends were broadly consistent with 2015. In the 2016 drug adverse reaction/incident report, 9.9% of children under the age of 14 were reported to be the same as in 2015. The number of elderly patients over age 65 was 23.5%, up 2.0 percentage points from 2015.
FIG. 5: the age distribution of adverse drug reactions/events in 2016
The adverse reaction/incident report of the drug involves the medical condition
According to the suspected drug category, 81.5% of chemical drugs, 16.9% of Chinese medicine and 1.6 percent of biological products (excluding vaccines) were in line with 2015.
Figure 6. Drug adverse reactions/event reports in 2016 relate to the distribution of drug classes
Delivery system in accordance with the drug statistics, 2016 report adverse drug reactions/events involving the dosage of drug distribution, intravenous drugs accounted for 59.7%, other injected (such as: intramuscular injection, subcutaneous injection, etc.) (3.4%), oral drug delivery system (33.7%), other (such as: for external use, patch, etc.) accounted for 3.2%. There was no significant change in the distribution of drug channels compared to 2015.
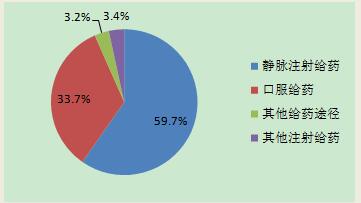 Figure 7. Drug adverse reactions/events reported in 2016
Drug adverse reactions/events, and the organ system
2016 reports of adverse drug reaction/event, involving the system in the top three for the skin and its accessories damage (27.6%), gastrointestinal system damage (25.4%) and systemic damage (10.9%). Chinese medicines, chemical medicine, involving the top three sorting system consistent with the overall situation, the top three biological products involving system and the overall situation is slightly different, in turn, is the skin and its accessories damage, systemic damage and immune dysfunction.
General case analysis of adverse reactions/incident reports
The overall picture of adverse drug reactions/events in 2016 has not changed significantly compared with 2015.
From adverse drug reactions/events report source, medical institutions accounted for 85.6%, compared with the 2015 report from the medical institutions increased by 3.4%, and the doctor report accounted for 55.5%, compared with 2015 increased by 1.5%, and suggests that medical institutions is still play a role of main channel of adverse drug reactions report; Number of reports from the pharmaceutical production enterprise flat compared with last year, the report number is still on the low side, carry out the work of adverse drug reactions monitoring showed that drug manufacturers hard enough; As a pharmaceutical production enterprise, should further strengthen their understanding of adverse drug reactions work, strengthen the listed drug safety study, establish risk management system, strengthen the awareness of corporate responsibility, risk prevention and control in time, give full play to the responsibility of the "drug safety first person".
Tips:
Is there an increase in the number of adverse drug reactions, the less safe the drugs?
The monitoring of adverse drug reaction is an important support for safety supervision after the market is listed, and its purpose is to discover and control the risk of drug safety in time. The measures for the administration of the adverse drug reaction report and monitoring stipulated in the state applies a system of adverse drug reactions report, the state encourages citizens, legal persons and other organizations report adverse drug reactions.
Adverse drug reactions report number, does not mean that the drug safety levels drop, which means that more and more comprehensive information we have, more understanding, the risks of drug risk more manageable, more basis to the evaluation of drugs, regulatory decisions more accurate. Similarly, in medical practice, to understand the performance of the adverse drug reactions occurred in a timely manner and degree, and maximize the avoided, also is the important measure to ensure medical safety.
(2) basic drug monitoring
National basic drug monitoring in general
2016 national adverse drug reaction monitoring network received national essential drug adverse reactions/events reported 595000 cases (accounting for 41.6% of the overall report), a 2.0% increase in 2015. There were 45, 000 serious reports, accounting for 7.5 per cent, up 1.1 per cent from 2015. The report covers 83.4 percent of cases of chemical and biological cases, and 16.6 percent of cases reported in China.
Analysis of chemical and biological products of national basic medicines
The catalogue of national basic drugs (grassroots medical institutions with using partial) "(2012 edition), chemical medicines and biological products, divided into 25 categories, 317 varieties. 2016 national adverse drug reaction monitoring network received 356 of the specific varieties, 502000 cases of adverse reactions/events report time, serious report 43000 cases, accounting for 8.5%.
2016 national essential drugs chemical medicines and biological products report by category, report the number of the top five were anti-infective drugs, cardiovascular drugs, antineoplastic agent, the digestive system drugs, analgesic and antipyretic anti-inflammatory / / fights rheumatism gout drug resistance, accounting for 72.2% of the basic drugs chemical report.
2016 national essential drugs chemical medicines and biological products adverse reactions/events report, adverse drug reactions/events involving the system of the top five is the skin and its accessories damage (27.4%), gastrointestinal system damage (27.2%), systemic damage (9.7%), the central and peripheral nervous system damage (9.0%) and immune dysfunction and infection (4.0%); The first five adverse reactions were secondary and 77.4%.
Analysis of the status of drug in the national basic medicine
The catalogue of national basic drugs (grassroots health institutions with using partial) "(2012 edition), proprietary Chinese medicine section of internal medicine, surgical medicine, medicine of department of gynaecology, ophthalmic preparation, ent, orthopedics 6 categories a total of 203 varieties. The 2016 national center monitoring network received 99,000 reports of 203 related species, of which 6,857 were reported, and 6.9 percent were reported.
2016 national essential drugs proprietary Chinese medicine part six classes, total number of adverse drug reactions/events report from more to less, in turn, for doctors of internal medicine, orthopedics, gynecology preparation, otolaryngology medication, surgery, ophthalmology drug use. Including internal medicine report the total number of 85.7% of the overall report number, this may be related to internal medicine clinical usage, and the list of essential medicines in traditional Chinese medicine injections are internal medicine. Internal medicine in the top five were quyu agent, WenLi agent, agent begin to understand, clear heat, the centralizer, the five kinds of drug report 90.0% of the number of internal medicine report.
2016 national list of essential medicines, proprietary Chinese medicine part of adverse drug reactions/events report involving the system of the top three is the skin and its accessories damage (29.0%), gastrointestinal system damage (23.4%) and systemic damage (14.3%). Different dosage form report involvement in the system, injection adverse reactions/events involving system in the top three is the skin and its accessories damage (21.4%), systemic damage (12.7%), gastrointestinal system damage (6.5%), oral preparations involving system in the top three is gastrointestinal system damage (16.1%), skin and its accessories damage (5.1%), nervous system damage (2.6%).
The above monitoring data shows that the overall situation of basic drug monitoring in 2016 is basically stable.
(3) monitoring of chemicals and biological products
General situation
In the 2016 drug adverse reaction/incident report, there were a total of 157,000 suspected drugs, including 81.5% of chemicals and 1.6% of biological products. The 2016 serious adverse events/incident report involved 12.10 million suspected drugs, including 86.9 percent of chemicals and 1.8 percent of biological products.
It involves the patient
In 2016, the proportion of men and women in the study of chemical and biological adverse reactions/events was closer to 0.97:1, women slightly more than men. 2016 chemical/biological products (excluding vaccine) cases of adverse reactions/events report, reports of children under the age of 14 patients (9.9%), accounted for 23.4% of elderly patients over the age of 65 years report. The severe adverse events/incident reports in 2016 involved the patient's age distribution in general.
It involves medicine
Adverse drug reactions/events in 2016 reports of suspected drug, chemical case number of the top five categories of anti-infection drugs (43.9% of the total number of cases of chemicals), drug cardiovascular system (10.4%), analgesic (6.2%), electrolyte and acid-base balance and nutritional medicine (5.9%), tumor (5.8%).
2016 chemicals serious adverse drug reactions/events in the report, the most common drugs was anti-infection drugs, the proportion is 35.0%, 0.9% lower than in 2015. The second is antitumor drugs, which account for 22.8%, the same as in 2015. 2016 report adverse drug reactions/events involved in biological products, antitoxin and immune serum (28.5%), cell factor accounted for 23.2%, accounted for 4.9% of blood products, diagnostic use biological products accounted for 0.1%.
According to the dosage form, the dosage form was 64.9 percent and the oral preparation was 32.1 percent in the 2016 chemical adverse reactions/incident report. More than 90% of biological products are administered by injection.
Analyze the overall situation
There was no significant change in the number of chemical and biological adverse reactions/events reported in 2016 compared with 2015. Chemical drugs, anti-infective medicine report number is still in the lead, but the proportion is down nearly 1% in 2015, has been declining for six years, prompt the anti-infective drugs improved the safety of clinical use. Dosage formulations and distribution, chemical injection preparation proportion continues to increase, prompt clinical should continue to focus on the use of injection dosage form management and safety monitoring. In patients age distribution, the proportion of elderly patients with adverse reactions report still slowly increase trend, and the trend of population distribution in our country changes, safety usage tips should continue to focus on the elderly population.
(4) monitoring of traditional Chinese medicine
General situation
In the 2016 drug adverse reaction/incident report, there were about 157,000 cases of suspected drug and 16.6% of Chinese medicines. In 2016, the national drug adverse reaction monitoring network received a total of 102,000 cases of severe drug adverse reactions/events, and Chinese medicine accounted for 5.5 percent.
It involves the patient
The proportion of men and women reported in the 2016 drug adverse reaction/event report was close to 0.87:1. In the 2016 drug adverse reaction/incident report, the number of children under 14 years of age was 8.0%, and 24.7% of those aged 65 and older reported. The results of the 2016 serious adverse drug reaction/incident report are in line with the overall situation of the patient.
It involves medicine
2016 report adverse drug reactions/events involved in the number of proprietary Chinese medicine cases of suspected drug's top ten categories are respectively manage blood agent in promoting blood circulation to remove blood stasis drugs (29.7%), heat in the heat of poison (9.7%), benefit qi and nourishing Yin in medicine (8.5%), begin to understand the cool prescriptions (8.4%), nourish the symplectic nourish the cold medicine (6.0%), clearing damp heat dehumidification in medicine (5.0%), clearing damp wind to wet in medicine (3.2%), clear heat phlegm in the expectorant drugs (2.4%), boosting agent mend angry medicine (1.8%), the principle of blood in the agent of yiqi huoxue medicine (1.5%). Compared with 2015, the number of the reported cases of leprotic blood drug in leaching agent was in the top 10, and the other categories were unchanged from the previous 10 before the withdrawal of cough. In the 2016 Chinese medicine adverse reaction/incident report, the proportion of injection and oral preparation was 53.7% and 38.6% respectively. The top 10 categories in the list of cases of severe drug adverse reactions/incident reported in 2016 were basically in line with the overall situation of Chinese medicine.
2016 report adverse drug reactions/events according to the dosage drug distribution and intravenous administration (53.2%), other injected accounted for 0.6%, oral medicine accounted for 40.2%, the other method is occupied 6.0%, compared with 2015, the general method distribution has no obvious change. Serious adverse drug reactions/events in 2016, the report according to the dosage drug distribution intravenous administration (85.8%), other injected accounted for 0.9%, oral medicine accounted for 12.1%, the other method is occupied 1.2%, compared with 2015, the general method distribution has no obvious change.
Analyze the overall situation
The number of adverse reactions/events reported in 2016 was the same as in 2015. From the report on dosage forms and the way of medicine, the injection of traditional Chinese medicine is higher, and the risk of the safety of the drug needs to be continued. Look from drug category, is primarily concerned with promoting blood circulation to remove blood stasis, heat-clearing and detoxicating, nourishing Yin, cool open TCM injections, prompt should continue to pay attention to the above categories and drug risk, risk control measures in a timely manner.
Third, relevant risk control measures
According to a 2016 adverse drug reaction monitoring data and evaluation results, the food and drug supervision and management of the administration of found unsafe drugs take corresponding risk control measures in time, in order to ensure public safety.
(a), issued the adverse drug reactions information bulletin. 4, on the alert for single vidarabine phosphate for injection can cause serious adverse reactions and super indications associated risks, alert the faeries bone remains risk of liver damage caused by oral preparations, focus on testosterone drugs risk of cardiovascular events, alert to the new compound leaf slice of long-term, heavy use of, or merger with other drugs containing similar components used, may lead to severe drug eruption serious adverse reactions such as severe allergic reactions, such as drug safety risk in a timely manner.
(2) the organization of rosiglitazone and its compound preparations, anti-hepatitis-jaundice injection, compound amino acid injection (18 aa) and similar preparations such as 19 (classes) and drug instruction modified, perfect the warnings in the manual, adverse reactions, taboos and other relevant information.
(3) to revoke the approval documents of two varieties of phenyguaniguanide and ketone, based on the evaluation results.
(4) release the pharmacovigilance express 12 period, reported the manic and pp, products containing andrographolide, polyethylene glycol (peg) interferon alpha 2 a foreign drug safety information such as article 63, 68 (class) varieties.
Four theory
(1) anti-infective medicine
Anti-infective drugs refers to kill or inhibit various pathogenic microorganisms of drugs, including antibiotics, synthetic antimicrobial, antifungal, antiviral, anti mycobacterium, etc. A total of 518,000 adverse reactions/events were reported in the national drug adverse reaction monitoring network in 2016, of which 33,000 cases were reported and 6.5% were reported. The adverse reactions/incident reports of anti-infection drugs accounted for 36.2 percent of the overall report in 2016. The number of anti-infection drug reports rose 1.1 percent in 2016 compared with 2015, with a severe report of 18.6 percent. The severity of the report was a 1.0 percent increase from 2015 (5.5%).
Drug situation
Anti-infective drugs in 2016 the number of adverse drug reactions report/events in the top three is cephalosporin, quinolone, large ring lactone class, the top three varieties of levofloxacin, azithromycin, ceftriaxone, compared with 2015, ranking no change. The top three most serious reports were cephalosporins, quinolones, and anti-tuberculosis drugs, and the number of anti-tb drugs rose to no. 3 by 2015. The top three strains of the 2016 anti-infection drug adverse reactions/incident report were levofloxacin, ceftria and cefoperazone. That's basically the same as 2015.
Anti-infection adverse drug reactions/events in 2016 report, injection (78.3%), oral preparations accounted for 19.4%, other dosage forms 2.3%, compared with the overall report drug, injection rate on the high side, dosage form distribution almost the same as in 2015. Serious adverse reactions/events in the report report, injection (82.1%), oral preparations accounted for 17.3%, other dosage forms 0.6%, compared with the overall drug serious report injection rate on the high side, compared with 2015, injection dosage form has fallen by 0.6%, up 0.5% on oral dosage forms.
The system of the organ system
In the report on adverse reactions/events in 2016, the overall report and the reported adverse reactions/events of adverse reactions/events of the drugs were shown in figure 8. In contrast to the overall report on anti-infective drugs, the total body damage, immune dysfunction and the composition of the infection and respiratory system were significantly elevated. Anti-infective medicine oral preparations involving system in the whole report before three is gastrointestinal damage (43.0%), skin and its accessories damage (27.6%), nervous system damage (6.8%). The first three parts of the injection system were skin and accessory damage (43.7%), gastrointestinal damage (20.2%) and systemic damage (8.6%).
Anti-infective medicine oral preparations involving serious report system in the top three is liver damage (27.1%), skin and its accessories damage (21.1%), systemic damage (9.5%). Injection involving system in the top three is the skin and its accessories damage (22.5%), systemic damage (19.1%), immune dysfunction and infection (12.7%).
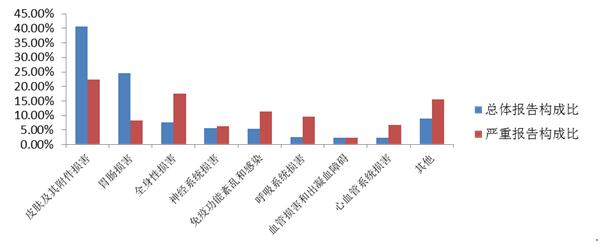 FIG. 8. Adverse reactions/events and organ systems in the anti-infective agent
Monitor situation analysis and safety analysis tips
2016 anti-infection drug adverse reactions/events report from 2015, the total has increased, serious report number increased by 18.6%, and growth level is lower than the overall case report growth in 2016. Compared with 2015, 2016, anti-infection/incident reports of adverse drug reactions accounted for the proportion of the overall report fell by 0.5%, the proportion of serious report decreased by 0.6%. Although anti-infection drugs in recent years, the report growth levels lower than the growth level of the overall report, but it still occupies the first place, its security has been focused on.
The highest proportion of quinolones in antiinfective drugs was second only to that of cephalosporins, with the severe adverse effects of fluoroquinolones. Adverse drug reaction monitoring network in our country by oral and injected dosage of fluoroquinolone adverse drug reaction performance involving system mainly involves the skin and its accessories damage (35.5%), gastrointestinal damage (27.3%), nervous system damage (8.5%), systemic damage (6.2%), immune dysfunction and infection (5.5%). Fluoroquinolone drugs may occur tendinitis and tendon lesion, deterioration of myasthenia gravis, peripheral neuropathy, severe arrhythmia, hypersensitivity, liver toxicity, central nervous system disease, blood disorders, photosensitive/light toxicity "and other serious adverse reactions.
The safety of fluoroquinolones has been closely watched, and new risks have been found. State food and drug supervision and management of administration since 2003 began to publish about the risk of fluoroquinolone drugs warning information, through multiple phase of the adverse drug reactions information bulletin warning of fluoroquinolone drugs serious adverse reaction stage (35, 58) and ciprofloxacin (5), and for the effect of stage (11, 24), levofloxacin (stage 22, 56), and other varieties of serious adverse reaction case, notification of adverse reactions related to systemic damage, nerve damage and mental system, the common serious adverse reactions, such as skin and its accessories damage also stressed the drugs to myasthenia gravis is aggravating, likely irreversible peripheral neuropathy, and affect the level of blood sugar control of diabetes mellitus and other risks.
Fluoroquinolone drugs more serious adverse reactions, and disabling of serious adverse events may occur, prompt clinical doctors prescription fluoroquinolone drugs to patients should weigh the pros and cons, while patients in the use of fluoroquinolone drugs should be carefully read product manual, if serious adverse reaction, appeared in the process of drug should be stopped immediately, and see the doctor as soon as possible.
Tips:
1, what are the fluoroquinolone drugs, which are taken by mouth and by injection?
Chinese listed by oral and injected to fluoroquinolone drugs have the effect of norfloxacin, pei fluorine, ofloxacin, levofloxacin, moxifloxacin, ciprofloxacin, maintain, fluorine Rosa star, in effect, the division of sand, kling of sand, for the effect of adding, reed fluorine, fluorine, fluorine effect, characters and characters and Jimmy effect.
What diseases are the main treatment for fluoroquinolones by oral and injection?
Fluoroquinolone drugs wide antimicrobial spectrum, especially for aerobic gram-negative bacteria have strong exterminate action, to gram-positive bacteria such as staphylococcus aureus, also has good antibacterial activity, some species of mycobacterium tuberculosis, mycoplasma, and is also effective in accordance with the original body and anaerobic bacteria. It is suitable for respiratory infection, urinary tract infection, gastrointestinal infection, joint and soft tissue infection.
What serious adverse reactions are caused by fluoroquinolones?
By oral and injected dosage of fluoroquinolone drugs can happen tendinitis and tendon lesion, deterioration of myasthenia gravis, peripheral neuropathy, severe arrhythmia, hypersensitivity, liver toxicity, central nervous system disease, blood disorders, photosensitive/light toxicity "and other serious adverse reactions. Some serious adverse events can lead to disability, such as the fluoroquinolone drugs caused by peripheral nerve lesions could be irreversible, after the use of fluoroquinolone drugs will happen soon, usually within a few days, in some patients, although have disabled fluoroquinolone, but the symptoms last more than one year. If the symptoms of patients with peripheral neuropathy, such as pain, burning, tingling, numbness, and/or weak, or other changes, such as light touch, pain, feeling, position, temperature and vibration feeling, etc., should be discontinued fluoroquinolone, replace with other non fluoroquinolone antibacterial drugs. Unless you continue to use the benefits outweigh the risks of fluoroquinolone treatment.
What should be noticed with fluoroquinolones?
By oral and injected to fluoroquinolone drugs more serious adverse reactions, and disabling of serious adverse events may occur, remind clinicians to patient prescription fluoroquinolone drugs should weigh the pros and cons, strictly grasp the indications, details about the use of the drug dosage, contraindications, precautions, adverse reactions, drug interactions, drug information such as the special crowd, make patients to obtain the best treatment effect, at the same time, try to reduce the risk of drug use. Patients in the use of fluoroquinolone drugs should be carefully read product manual, pay attention to such drugs warning, adverse reactions and points for attention, such as security information, if serious adverse reaction, appeared in the process of drug should be stopped immediately, and see the doctor as soon as possible.
(2) Chinese and western medicine compounds
The "herbal preparation" in this report refers to "the combination of traditional Chinese medicine, natural medicines and chemical medicines composed of traditional Chinese medicines, natural medicines and chemicals". Received 2016 national adverse drug reaction monitoring network of Chinese and western medicine compound preparation of adverse drug reactions/events reported 13000 cases (accounting for 0.52% of the Chinese traditional medicine adverse reactions/events report), among which 4437 were new, serious report, accounting for 34.2%.
Patient condition
In the adverse reaction/incident report of the drugs of Chinese and western medicine, 51.6% of men and 48.3% of women were women, and no significant gender differences were observed. Chinese and western medicine compound preparations of serious adverse drug reaction/event report, male patients (35.3%), female patients (62.9%), the structure of the female patients is relatively high.
Chinese and compound preparations of serious adverse drug reaction/event report, the composition of 65 and older patients than higher than its constituent ratio in the adverse drug reactions/events report, the composition of 15 to 44 patients less than its constituent ratio in the adverse drug reactions/events report. As shown in figure 9.
 The age composition of the drug adverse reactions/events reported in the Chinese western medicine
Drug situation
In 2016, 16 of the top 20 drugs for drug adverse reactions/incident reports were oral preparations and four were external agents. Eight of the top 10 drug reagents for severe drug adverse reactions/incident report were oral preparations, and two were external agents.
The system of the organ system
In 2016, Chinese and western medicine compound preparations of adverse drug reactions/events report involving organ system shown in figure 10, serious adverse drug reactions/events report involving organ system is shown in figure 11.
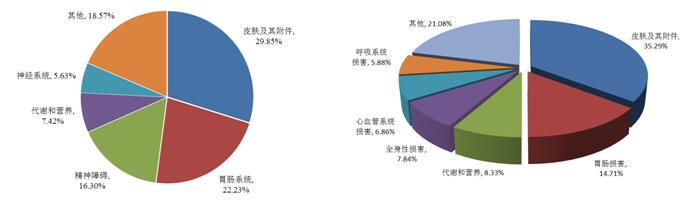 FIG. 10 the adverse reaction/incident report of the drug in the system of the organ system
Monitor situation analysis and safety analysis tips
Chinese and western medicine compound preparations, in addition to the traditional Chinese medicine (TCM) still contains one or more ingredients, such as antipyretic analgesics (acetaminophen, salicylic acid, indomethacin, etc.), cough/expectorant antiasthmatic (cough, hydrochloric acid by oprah winfrey ketone ketone new chlorine, hydrochloric acid, ammonium chloride, hydrochloric acid, bromine has asthma, etc.), anti-infection drugs (levamisole hydrochloride, berberine hydrochloride, nitrofural, furazolidone, sulfanilamide armour oxygen pyrimidine, etc.), hypoglycemic/blood pressure drugs (glyburide, clonidine hydrochloride, etc.), antiallergic (maleic acid chlorobenzene that sensitive, chlorine, hydrochloric acid to hydroxyl lamictal, etc.), vitamins (vitamin A, B1, B2, B6, C and D2, etc.) and amino acid (lysine, leucine, threonine, etc.), local anesthetics (hydrochloric acid cloth than paid, procaine hydrochloride, etc.), etc.
Part of Chinese and western medicine compound preparations in clinical commonly used ones are, as the same as the other contains ingredients or efficacy similar drug combination, the superposition due to dose or effect, could lead to a drug overdose or toxicity of synergy. Monitoring data show that, compared with alone (13.0%), Chinese and western medicine combination drug combination involving the gastrointestinal system of serious adverse drug reaction/event is higher (20.0%). May be related to individual patients in the use of Chinese and western medicine of compound preparation at the same time, taking other medicines containing effect the same or similar components, resulting in dose or superposition of effect factors.
Administration in recent years on the drug release harmful response information bulletin, prompt the Jane chrysanthemum antihypertension tablet, clear cold tablets (capsules) and brain collaterals capsule, a new compound leaf slice of Chinese and western medicine compound preparations such as clinical application security risks; At the same time, the paper also revised the instruction of several Chinese and western medicines, such as the new compound leaf blade, to improve the safety and risk information of the drug, and to guide the use of clinical safety.
Tips:
How do you safely use the Chinese medicine?
Note the safety of the components of the Chinese and western medicine: the use of hydrochlorothiazide, which may lead to hypokalemia in the long term, is the result of the use of the product. The new compound, which contains isoprene, may be used for long-term use.
2, avoid drug overdose: Chinese and western medicine compound preparation and other contain the same ingredients or efficacy similar drugs used in combination, easy to cause some formula composition of ultra dose use or cause toxic synergy. When used, the combination of the same components or the effect of the effect of the same components should be taken into account to avoid the occurrence of drug overdose or toxic synergies.
3, reduce drug interactions: drug use should be paid attention to when Chinese and western medicine compound preparations of interaction between components and other drugs, dosage should be adjusted when necessary or replace other drugs, in order to reduce or control for drug interactions cause security risk.
(3) antiinflammatory drugs for relieving fever
It is also called non-steroidal anti-inflammatory drug (NSAIDs), which is a type of drug that can relieve heat, pain, anti-inflammatory (aseptic inflammation) and rheumatism. In 2016 the national adverse drug reaction monitoring network received antipyretic analgesic anti-inflammatory drug (including the antipyretic analgesic anti-inflammatory drug monotherapy and compound preparations) of 62323 cases of adverse reactions/events report, accounting for 5.2% of the overall report for chemical medicine. Severe adverse reactions/events reported 2,951 cases, which accounted for 4.7% of the total reported number of antiinflammatory drugs.
Drug situation
2016 antipyretic analgesic anti-inflammatory drug adverse reactions/events report, on aniline, propionic acid, destroy acid, salicylic acid, enol acids (yesterday kang class), selective cox-2 depressants, pyrazole ketones, indoles and other nine categories of drugs (as shown in figure 12), including a large proportion of compound preparations, such as ammonia coffee huang tablet, compound ammonia Lin barbital injection and compound amantadine capsule, etc.
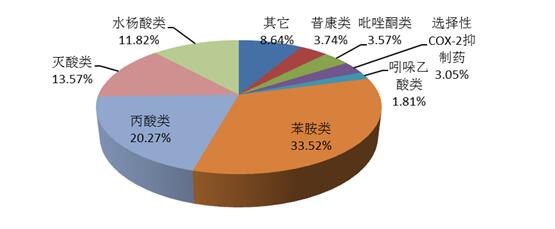 FIG. 12 categories and number of reports of dethermal analgesia in 2016
In the report on the adverse reactions/events of the 2016 anti-fever anti-inflammatory drugs, the injection rate was 79.9%, higher than the total number of injections (64.9 percent) in the overall chemical report. Oral preparations accounted for 12.7% of the total, lower than the proportion of oral preparations (32.1%) in the overall report.
The system of the organ system
2016 antipyretic analgesic anti-inflammatory drug adverse reactions/events report, the affected organ system in the top three in turn for gastrointestinal damage (44.8%), skin and its accessories damage (20.1%), nervous system damage (10.2%), as shown in figure 13. The proportion of gastrointestinal damage (25.4% overall) was significantly higher compared to the overall drug report.
.jpg) FIG. 13: the distribution of anti-inflammatories and organ systems in 2016
In antipyretic analgesic anti-inflammatory drug serious adverse reactions/events in the report, the oral agents involving organ system in the top three in turn for gastrointestinal damage (34.7%), skin and its accessories damage (19.0%), liver damage (6.0%). Injection serious report involving organ system in the top three followed by systemic damage (22.7%), skin and its accessories damage (18.7%), respiratory damage (15.0%).
Monitor situation analysis and safety analysis tips
The outstanding characteristics of the adverse reaction of the antiinflammatory drugs were the gastrointestinal damage, up to 44.8%, which was 19.4 percentage points higher than the total gastrointestinal damage in the database. Gastrointestinal damage is the most common antipyretic analgesic anti-inflammatory drug adverse reactions, especially long-term or large doses used, may lead to gastric ulcer, perforation and bleeding, especially in the majority with cases of gastrointestinal bleeding caused by aspirin, should be cause for concern.
The proportion of injection preparation is higher than the total dose of the drug in the antiinflammatory drug. The number of injectables in the first two positions was a short-term withdrawal, with a significantly higher proportion of children under 14. Serious adverse reaction is mainly anaphylactic shock, allergic reactions, severe skin rashes, breathing difficulties, including anaphylactic shock, especially in the majority with young group.
There are a wide variety of compound agents for antiphlogistic drugs. In addition to different antipyretic analgesics into nowhere, may also jointly with the other kind of drug ingredients prescription, used to relieve symptoms such as pain or cold, clinical dosage is bigger, the occurrence of adverse reaction is relatively more. In oral reagents, the number of cold medicines was at the forefront, and severe adverse reactions were mainly caused by allergic reactions and rashes. Some patients may have long-term or overdose of antiinflammatories, which may cause severe liver and kidney damage. Therefore, the use of such drugs should be weighed the advantages and disadvantages, especially for the common cold, fever, the treatment of mild pain, grasp the rational drug use common sense, or under the guidance of doctors and pharmacists and medical treatment.
Tips:
Do you have to take an antipotion for fever?
The antipyretics are only treatment for disease, and fever is used to reduce or restore body temperature to normal levels. On the other hand, fever is not a bad thing; it is a defensive reaction of the body. When the body's phagocytosis increases, white blood cells increase, and the number of antibodies increases, which is good for killing bacteria and viruses. So, cold fever in adults, such as the high fever (more than 38.5 ℃), generally do not advocate using antipyretic; The fever should be admitted in time to the doctor or pharmacist. Some used in antipyretic antipyretic analgesics, especially injection (antifebrile injection), may cause severe allergic reactions, such as severe rash, anaphylactic shock, so don't cry because it is the pursuit of antifebrile effect and speed, and blindly use antifebrile injection.
Is there a risk in using anti-cold medicine?
Cold is mostly consists of several components of compound preparations, common ingredients are acetaminophen, aspirin, diclofenac, ibuprofen, chlorobenzene that susceptibility, diphenhydramine, caffeine, etc. The drugs themselves can cause adverse reactions, such as rashes, drowsiness, and gastrointestinal reactions. A handful of patients may also be a serious adverse reactions, such as the use of acetaminophen can cause serious liver damage, bullous skin rash, etc., using GanMaoTong hematuria (especially children). In addition, cold is more compound preparations, used to avoid contain the same ingredients of different drugs used in combination, this kind of use increases the risk of the security of the cold.
How to prevent and reduce the gastrointestinal bleeding caused by aspirin?
Gastrointestinal bleeding is a common and severe adverse reaction of the anti-inflammatory drugs. Aspirin, for example, can affect the function of platelets, making the blood less resistant to coagulation and causing bleeding. Clinical use of aspirin should be cautious of patients have the following factors: has a history of peptic ulcer, hemorrhagic disease, patients with severe liver disease, old age, long-term or large dose of medication, combined use of anticoagulants (such as law that China, clopidogrel), etc. Postprandial use can reduce the stimulation of aspirin on gastrointestinal tract, combined use of proton pump inhibitors in accordance with the doctor's advice when necessary (antacids) or gastric mucosa protectant, reduce the occurrence of gastrointestinal bleeding. If the patient has the symptom such as stomach, oil, bleeding from the gums, bleeding from the nose, the patient should be admitted in time.
Iv. The treatment of mental disorders
Mental disorders in the adverse drug reaction monitoring network medications including antipsychotics, antidepressants, manic drug resistance, anxiolytics, psychostimulant drugs 5, 2016 national adverse drug reaction monitoring network received mental disorder treatment drug adverse reactions/events reported 25000 cases, accounting for 1.73% of the overall report 2016 number; Of these, 2714 were reported, accounting for 2.65 percent of the total serious report, and 10.9 percent of the adverse reactions/events reported by psychiatric disorders.
Patient condition
Mental disorder treatment medicine in 2016 cases of adverse drug reactions/events report, male 13000 cases, 12000 cases of female gender is unknown in 78 cases, male proportion about 5.8% than women; Severe cases were reported in 1450 men, 1,246 women and an unknown number of 18 cases, with severe cases reporting a 7.5% higher proportion of men than women.
Mental disorders in 2016 reports of adverse drug reactions/events for the treatment of drug age distribution overall, aged 15 to 44 (55.0%), 45-64 age group (32.6%), 65 years of age or older age group (11.1%), the proportion of patients with the rest of all ages were lower than 1%. The age distribution of the severe report was similar to the overall age distribution for psychiatric disorders.
Variety situation
Mental disorder treatment drug adverse reactions/events in 2016 report on 5 species, 27000 cases (3136 cases), including antipsychotics 73.1% 73.3% (severe), antidepressants accounted for 21.5% 20.8% (severe), anxiolytics 3.9% 4.4% (severe).
2016 mental disorder treatment drug adverse drug reactions/events report, injection preparations accounted for 7.6%, oral preparations accounted for 92.1%, 0.3% other dosage forms, oral dosage forms of significantly higher than the national adverse drug reaction monitoring overall, the report of the oral dosage forms of database may be related to mental disorder treatment of drug dosage form is given priority to with oral dosage forms.
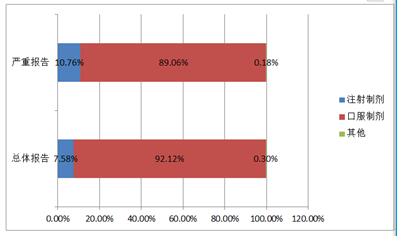 Figure 14 drug adverse reactions/event reporting dosage distribution for mental disorders therapy
The cumulative organ system
2016 spirit system, drug adverse reactions/events report adverse drug reactions/events involving system is the top five of the nervous system damage (33.7%), gastrointestinal damage (18.2%), cardiovascular system damage (10.3%), mental disorders (9.5%) and liver damage (7.8%). The secondary and 79.4 percent of the adverse reactions in the first five levels of the system were followed. Drug treatment of mental disorder serious adverse drug reaction/event report, involving the system in the top five were nervous system damage (28.6%), liver damage (17.9%), cardiovascular system damage (14.0%), gastrointestinal damage (8.0%), the blood system damage (7.7%).
Monitor situation analysis and safety analysis tips
This year's report psychiatric disorders medications including antipsychotics, antidepressants, manic drug resistance, anxiolytics, psychostimulant 5 classes, from the mental disorders involving drugs, drug adverse reactions report antipsychotic drugs report number and the quantity of serious report are among the top of the various kinds of drug treatment for mental disorders and mental disorder treatment medicine reported 73.1% respectively, and 73.3% of severe mental disorder treatment medicine report. Most of these cases are atypical antipsychotics. The safety of such drugs should be fully understood, and the clinical and rational use of drugs should be guided to reduce the risk of drug use.
Tips:
What is atypical antipsychotics?
Atypical antipsychotics are mainly used for acute and chronic schizophrenia and other various psychoticism state positive symptoms and the treatment of negative symptoms, compared with the traditional antipsychotic drugs and atypical antipsychotic drugs have stronger antipsychotic effect and less of extrapyramidal reactions, thus more widely applied in clinical. At present, the listing of the atypical antipsychotics include: clozapine and risperidone, olanzapine, quetiapine, ziprasidone, o pp, pp luo long horse, peary pp ketone, tolbutamide will, etc. Atypical antipsychotic drugs, mainly for the central nervous system reaction such as dizziness, drowsiness, insomnia, epilepsy, extrapyramidal reactions, agitation, mania, delirium, etc. In addition, there are some severe adverse reactions, such as granulocytosis and glycolipid metabolism.
What should paramedics be aware of in the use of atypical antipsychotics?
In the case of severe adverse reactions to atypical antipsychotics and their risk factors, medical personnel should fully recognize and pay attention to them. In the case of prescription drugs, the doctor should carefully inquire the patient's past history and should strengthen the monitoring and monitoring of the patient during use. Should communicate with the patient and their families or guardians the risk of the drug, and instruct the patient to use the drug correctly and rationally.
How do patients correctly use atypical antipsychotics?
The patient should follow the advice of the prescription doctor and follow the prescribed atypical antipsychotics. Read the instructions in detail to understand the adverse reactions and precautions. This class of drugs to use for a long time, because for a doctor to ask for the regular monitoring of laboratory tests should fully understand and actively cooperate with, medication after experiencing discomfort symptoms should be timely.
(5) children's medicine
Children's drug use refers to the use of special medicines for minors under the age of 14, and the children's drug report refers to the report that the patient is 14 years old.
2016 national adverse drug reaction monitoring network received report from medical institutions, 1.2 million children aged 0 to 14 patients related to the adverse drug reactions/events report 128000, accounting for 10.6%, compared with 2015, down 0.2 percentage points. A total of 6,986 children were reported from medical institutions in 2016, 5.5 percent of the total number of children reported, up 0.4 percentage points from 2015.
Variety situation
2016 children adverse drug reaction/event report, the chemical medicine accounted for 84.0%, which accounted for the top three, respectively, anti-infection drugs (76.5%), electrolyte and acid-base balance (5.9%), respiratory medicine and nutrition medicine accounted for 5.1%; Chinese medicine accounted for 13.5 percent of the total, accounting for 36.6 percent of the total, 18.2 percent of the disagent, and 17.7 percent for the opening. Biological products account for 0.9%. In the 2016 children's severe drug adverse reactions/incident report, the chemical medicine accounted for 87.3 percent, Chinese medicine accounting for 10.4 percent, biological products 1.4 percent, and unknown 0.8 percent.
In the 2016 children's drug adverse reactions/event report, the injection was 84.9 percent, oral preparation 11.8 percent, and other preparations accounting for 2.6 percent. The proportion of injection and oral preparation was 86.5% and 10.3%, respectively, in the adverse reaction/incident report. In the adverse reaction/incident report, the proportion of injection and oral preparation was 74.7% and 22.2% respectively. 2016 children serious adverse drug reactions/events involved in the distribution of pharmaceutical dosage forms, injection (90.8%), oral preparations accounted for 6.8%, other preparations accounted for 1.9%, 0.5% is unknown.
The cumulative organ system
2016 children adverse drug reactions/events in the report, the overall report and serious adverse drug reactions/events the cumulative organ systems as shown in the figure 15 and figure 16. The top three were skin and accessory damage (52.5%), gastrointestinal damage (20.5%) and systemic damage (8.3%). The three Chinese medicines, chemical medicine, involving system consistent with the overall, involving the system of biological products and the overall sorting, respectively is systemic damage, skin and its accessories damage, administration site damage to give priority to. 2016 children serious adverse drug reactions/events report, involving the system of the top three is the skin and its accessories damage (27.6%), systemic damage (23.0%), respiratory damage (8.7%).
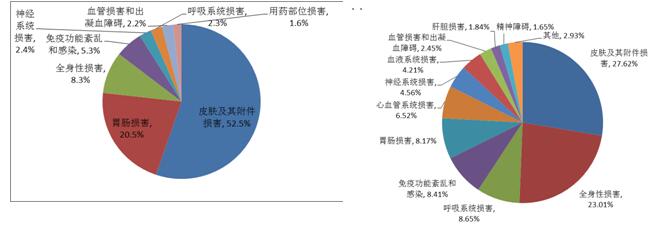 FIG. 15. The adverse reaction/incident report of the drug and the system of the organ system
Monitor situation analysis and safety analysis tips
The number of children aged 0-14 in 2016 was 10.6% of the total reported by medical institutions, down from 2015. One serious report, 5.5% of children report children patients involved in the proportion of new general and serious report is below is from the report the overall situation of medical institutions.
The number of suspected drugs involved in children's drugs was mainly chemical, accounting for 84.0%. Involves dominated by injection of drug dosage forms and general report and severe accounted for 84.9% and 84.9% respectively, compared with 2015, decreased by 0.8% and 0.8%, respectively, show that reasonable use to further strengthen injection in children. Injection in the use of children, is associated with poor oral medication adherence in children, but children as a relatively special group, because the body organs and the physiological function and development of organizational structure is not fully, low immunity, or inadequate or lack of drug metabolism enzymes, plasma protein binding ability is poor, poor of water, electrolyte metabolism and so on drug absorption, distribution, metabolism and excretion, unlike adults, such as for drug sensitivity and tolerance differ with adult far, for children to use injected risk is bigger also, so prompt clinical medication should understand the physiological and pathological characteristics of child development period and the physical features, under the guidance of, and the rational use of drugs, can choose oral, topical medications were given at the same time do not choose injected.
Tips:
Children's common diseases and medication precautions
Children with fever: (1) temperature around 38 ℃. Take antipyretic analgesic drugs at the same time, to follow the doctor's advice, the share of antibiotics and antiviral drugs, under the guidance of the routine urinalysis performed inspection, to determine is a bacteria or virus infection. Antipyretic should drink plenty of water at the same time, promote the body heat and cooling, and avoid the continuous repetition medication or drug overdosage and lead to excessive sweating, the body temperature drop too fast caused by collapse, water and electrolyte balance disorder.
(2) diarrheal: the world health organization stipulates that every day a loose stool is equal to or more than three times, whether or not there is mucus, called diarrhea. Due to improper feeding, or other causes of diarrhea, taking antidiarrheal at the same time, under the guidance, take antibiotics or use of oral rehydration salts (ors) to prevent dehydration.
Set up and develop the correct behavior habits of children's drug safety
(1) safe dose: develop the habit of reading instructions. When giving medicine to your child, it is advisable to write the correct usage and dosage in clear and large words.
(2) safety handling: the habit of establishing a drug treatment to be placed in a safe place for children to be safely accepted. This behavior will prevent you and others from taking risks and protecting the environment.
(3) medication under the guidance of a doctor.
Relevant explanation
(1) this year's report is based on data from the national drug adverse response monitoring database on January 1, 2016, which is reported in the region on December 31, 2016.
(2) as in most countries, our country adverse drug reactions reports is through spontaneous reporting system to collect and input to the database, there are limitations of spontaneous reporting system, such as omission, fill in is not perfect, the information, can't calculate the incidence of adverse reactions, etc.
(3) the number of each type of adverse drug reactions/events report by the drug usage and the incidence of adverse drug reactions, and many other factors, so the adverse drug reactions/events report quantity does not directly represent the discretion of the incidence of adverse drug reactions or severity.
(4) the annual report is complete, some serious report, death is still in the process of investigation and evaluation, all the results are the real reflection of data collection situation, some problems do not represent the final evaluation results.
(5) professional analysis, the correlation of drug and adverse drug reactions/events to extract the drug safety risk information, according to the universality of the risk or severity, decide whether to take relevant measures, such as in the leaflet to join the security information, update how to safely use drug information, etc. In rare cases, drugs will be removed when the benefits of drugs no longer outweigh the risks.
(6) this year's report does not contain the monitoring data for the adverse reactions/events of the vaccine.
|
