Medical network on April 27 - recently, people club department issued "about 2017 national basic medical insurance, inductrial injury insurance and maternity insurance drug catalogue negotiations range of circular, involved in clinical use are the main drugs of major diseases such as cancer, cardiovascular disease, leukemia drugs high-end varieties, including 24 kinds of imported drugs, 20 kinds of domestic new, focused on innovative drug, exclusive listed in the category, including six localization of medicine is the 12th five-year plan approved 1 class innovation.
Anti-tumor targeted drugs are rapidly democratized
In the health care negotiation drug, the tumor immune-targeting drug takes up half of jiangshan, including monoclonal antibodies and small molecular-targeted drugs.During the 13th five-year plan period, China's main market for anti-tumor immunotherapy will enter the era of targeted therapy.
According to m Intranet HDM system data, 2015 domestic cities, public hospitals, public hospitals at the county level, the urban community health care, in towns and townships, entity drug stores, shop three terminal 6 big market antitumor immune market size is 95 billion yuan, is expected to amount to 109.8 billion yuan, 2016 year-on-year growth rate of 15.57% in the previous year.
According to the HDM system data, in 2015, the health talks involving 20 antitumor immune targeted drugs at home three terminal 6 large sum of money market is 12.8 billion yuan, including urban public hospitals accounted for 31.05%, to $3.975 billion.The 20 drugs were projected to contribute 45.46 billion yuan to the urban public hospital market in 2016, up 14.36 percent year on year.
The medicare drug involved in negotiations of 20 tumor immune targeted therapy drugs including nine biological engineering, rituxan, by bead resistance, beacizumab bead sheet resistance, this bead sheet resistance, cetuximab and human recombinant endostatin, and licensed resistance, compaq heap, hideo xidan resistance.The other is a small molecular targeted drugs such as sorafenib, it for, the path for, lapa for Nepal, paclitaxel liposome, west of the amine, according to the dimension of therapy and boron for with rice, fluorine d SiQun, and drug lenalidomide abiraterone.
Species trap
Taxol: the largest variety of cancer drugs in China
Taxol is a broad-spectrum antitumor drug, because of its unique mechanism of action, it is suitable for almost all solid tumor, now is a line, breast cancer, ovarian cancer treatment for lung cancer, colorectal cancer, melanoma, head and neck cancer, lymphoma, brain tumors are showing activity, is the largest varieties of cancer drug market in China.
Taxol was first discovered in the 1960s and is still widely used clinically.Taxol is also a "double-edged sword", in its composite under the influence of solvent, the allergic reaction, toxic renal damage, nerve toxicity and cardiovascular toxicity, caused paclitaxel (taxol) back the black box warnings.The breakthrough was achieved in clinical treatment after the introduction of albumin with paclitaxel and paclitaxel.
The Chinese taxol lipid body was listed in 2003 by nanjing green leaf pharmaceutical, which is called "lipoplin".So far the CFDA has approved 40 domestic enterprises producing Taxol injection, imported products with bristol-myers squibb Taxol (Taxol), Australian ding Anzatax (Anzatax), a new American biotechnology company type injection combined with albumin KaiSu Taxol (Abraxane).
According to HDM system data, 1-3 quarter of 2016 key cities for the domestic public hospital drug taxol amount to 1.015 billion yuan, predict drug taxol amount to 1.395 billion yuan in 2016, year-on-year growth of 13.32% last year, the market prospects.Domestic paclitaxel injection (16.47%), imported and paclitaxel injection (13.80%), the new type albumin in combination with paclitaxel KaiSu biological company occupy 9%, nanjing green leaves on the force occupy more than 60% of the market, the overall market has more than 2.5 billion yuan.At present, nanjing pharmaceutical leaves flapping element and the force of domestic paclitaxel injection average spread around five times, and significantly lower adverse reactions and side effects, which is an advantage in the market.
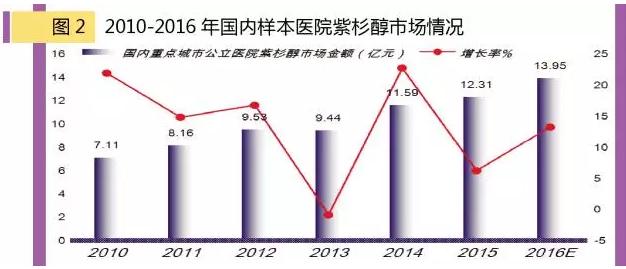
Be worth what carry is, once the paclitaxel liposome listed in the catalog of national health insurance program on negotiation success, will overturn paclitaxel injection market, market reshuffle is inevitable, decline of paclitaxel injection market worse, or will gradually fade out the market.On the other hand, it will promote domestic paclitaxel injection combined with albumin type product launches, domestic shiyao, hengrui, qilu (hainan), zhengda companies such as the weather is fine, has been made for testing and also developed a polymer micelle agent, micro emulsion, a new dosage form lipid microsphere agents etc.
Trastuzumab: the domestic market is more than 1.6 billion yuan
Trastuzumab (herceptin) is the recombinant DNA derived monoclonal antibody.On September 25, 1998, the U.S. FDA approved trastuzumab, which is called Herceptin.The European Union has approved herceptin as a first-line drug for human epidermal growth factor receptor 2 (HER2) positive metastatic breast cancer.In 2002, the CFDA approved roche's trastuzumab, which is called herceptin, for HER2 positive advanced breast cancer treatment, and is now a domestic exclusive.Herceptin targeted biological gene therapy with the ordinary compared radiotherapy, chemotherapy, hormone therapy, breast cancer, can be targeted killing tumor cells through gene selection, and does not affect the survival of human normal tissue cells.
According to the HDM system data, 1-3 quarter of 2016 key cities for the domestic public hospital drug herceptin amount to 441 million yuan, predicted in 2016 major cities in our country public hospital drug herceptin amount of 588 million yuan, year-on-year growth of 14.41% a year.The domestic market was 16.33 billion yuan.
Bevacizumab: the domestic market is more than 1.8 billion yuan
Bevacizumab is a metastatic colorectal cancer drug approved by the us FDA in 2004. The product is called Avastin.With the broadening of clinical indications, it is now approved for indications such as non-small-cell lung cancer, renal cell carcinoma, colorectal cancer and glioblastoma.At the end of 2012, the European Union approved its use for recurrent ovarian cancer.Bevacizumab has been listed in more than 120 countries worldwide and is a blockbuster drug for antibody.
In 2010, the CFDA approved roche's bevacizumab registration for metastatic colorectal cancer, which is called "avvitine" and is a domestic exclusive.Bevacizumab is of the biggest highlights of the fundamental changed the way to the role of tumor cells, no longer acting on tumor cells, but the effects on surrounding the tumor microenvironment, cut off the blood supply to the tumor area, on the proliferation of velocity anomaly tumor blood vessels.Global bevacizumab sales in 2016 reached $67.83 billion.
According to HDM system data, 1-3 quarter of 2016 key cities for the domestic public hospital drug avastin amount to 262 million yuan, to predict the sum of 349 million yuan in 2016, year-on-year growth of 34.69% a year.The domestic market is more than 1.8 billion yuan, accounting for 13 percent of the 20 tumor targets.
Recombinant vasopressin: the sample hospital was over 100 million yuan
Domestic another commendable antitumor biological targeted drugs is human recombinant endostatin (en), is a country with a simcere r&d innovation class of drugs.Grace degree combined standard chemotherapy scheme can improve the advanced non-small cell lung cancer patients with a median survival time and total survival rate, improve the patients with advanced non-small cell lung cancer chemotherapy in the clinical efficient and the rate of clinical benefit, are effective in patients with squamous carcinoma and adenocarcinoma.
Endu was a pioneer of the acquisition of the products in the arms of shandong madejin, which was listed and made famous in 2006.At present, the pharma industry has stepped up research on the research on the treatment of the encal-reducing agent, freeze-dried powder, new indication, new drug treatment, etc.
According to HDM system data, 1-3 quarter of 2016 key cities for the domestic public hospital er degree sum of nearly one hundred million yuan, predicted in 2016 major cities in our country public hospital degree is the sum of 111 million yuan, year-on-year growth of 16.48% a year.
Nitozumab: the growth rate is significant
Nitozumab (tyxin) is the first humanized antibody to treat solid tumors in China.2005 Beijing hundred Thai biological pharmaceutical industry to obtain new drug certificate, 2007 public permission, and in 2008 was rated as ten big popular drugs, is the world's third monoclonal antibodies for the treatment of solid tumor.
Tai xin has targeted strong, high specificity and the characteristics of low toxicity, put, chemotherapy can enhance the efficacy of treatment, used in the treatment of epidermal growth factor receptor expression Ⅲ/Ⅳ period nasopharyngeal carcinoma, for head and neck tumor, glioma, colorectal cancer, pancreatic cancer, non-small cell lung cancer and other cancer curative effect is distinct, little adverse reaction, which has a high clinical value.
According to HDM system data, 1-3 quarter of 2016 key cities for the domestic public hospitals, this bead single drug resistance to amount to 114 million yuan, predicted in 2016 key cities in our country public hospital, bead sheet resistance to the sum of 157 million yuan, a year-on-year growth of 21.88% a year.
|
